The FDA’s Commissioner’s National Priority Voucher (CNPV) Pilot Program, launched in June 2025, creates a highly accelerated 1–2 month review pathway for products addressing key national health priorities such as unmet medical needs, domestic manufacturing resiliency, public health threats, healthcare affordability, and advanced therapeutic innovation. Entry into the program may occur through FDA nomination or a brief sponsor- submitted application.
White Paper
Commissioner’s National Priority Voucher (CNPV) Pilot Program

Related Resources
All Resources
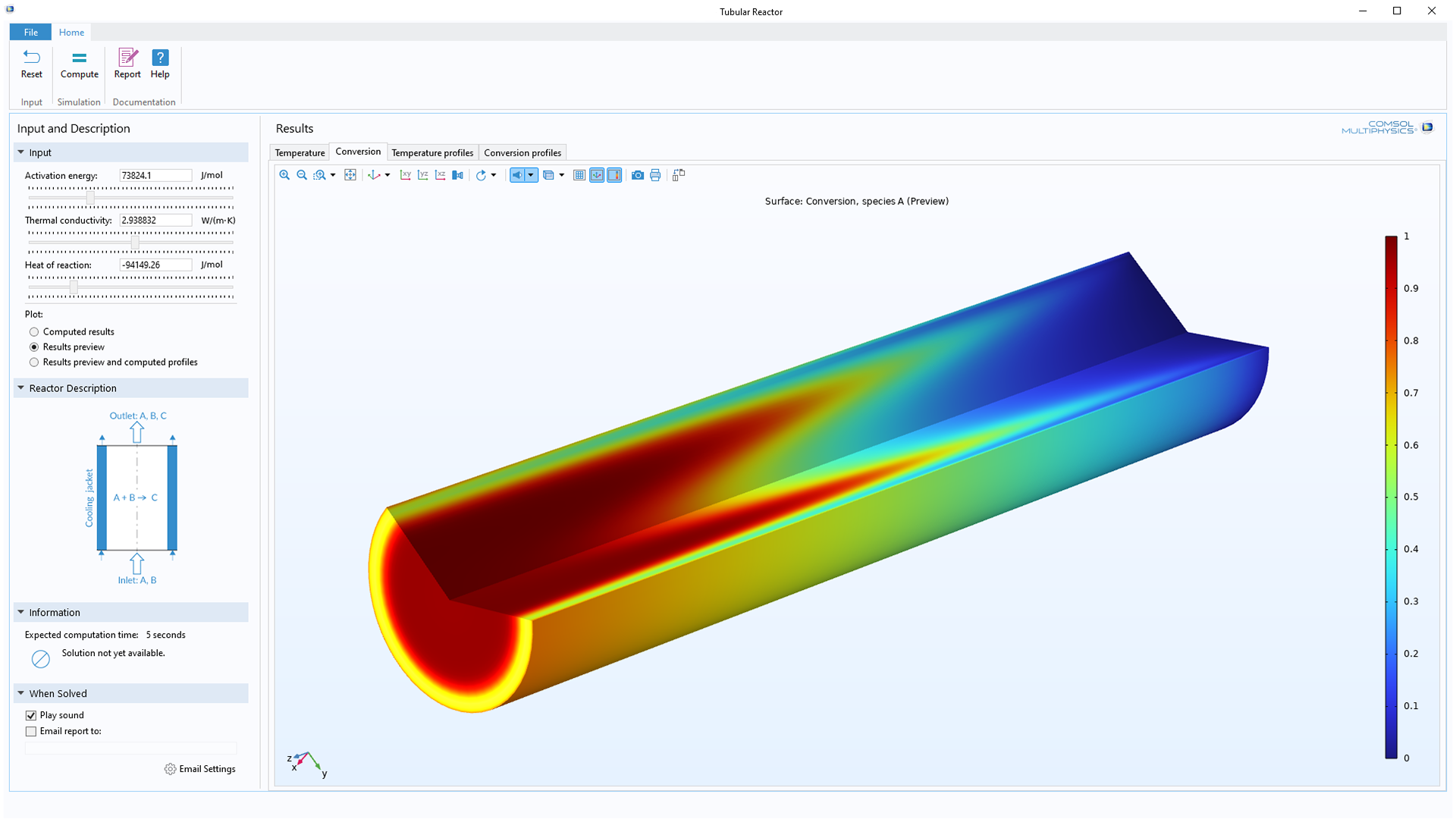
post
CFD Applications in Design of Manufacturing Processes and Facilities
Read More

post
How Regulatory Strategy Drives Innovation in Biopharma
Read More

post
Why Early-Stage Regulatory Planning is Essential for Biopharma Success
Read More

post
The Path to Breakthrough Therapies: A Comprehensive Guide to Drug Development
Read More
Let's move from science to success.
Let’s Talk