Cracking the Code: Syner-G’s Blueprint for Success with Pharmaceutical Stability Studies
Every year, the FDA handles a staggering 1,500 Investigational New Drug (IND) applications, a critical step in bringing pharmaceutical innovations to market. The IND review process is pivotal, ensuring the safety of study subjects and the integrity of clinical trials. To successfully navigate this complex landscape, understanding the FDA’s priorities, common stumbling blocks, and the benefits of pre-IND meetings is essential.
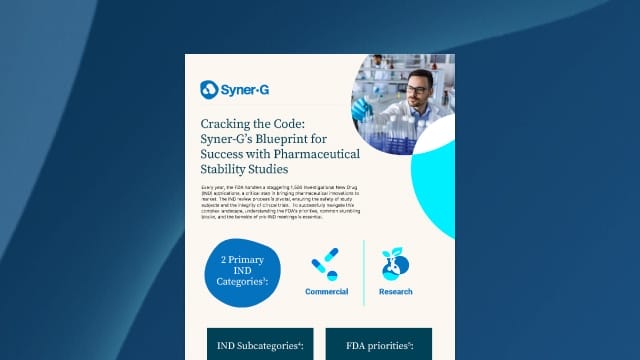
2 Primary IND Categories:
• Commercial.
• Research.
Research IND Types:
• Investigator.
• Emergency Use.
• Treatment.
• Exploratory.
FDA priorities:
• Animal Pharmacology and Toxicology Studies.
• Manufacturing Information.
• Clinical Information Including Protocols.
Common stumbling blocks:
• Insufficient nonclinical data.
• Insufficient safety, pharmacodynamic, and/or pharmacokinetic information.
• Inadequate information around manufacturing processes, control measures, and product characterization.
• Weak trial design.
• Inadequate regulatory documentation.
Pre-IND meetings:
• Proactively prevent potential issues.
• Provide insights into preparation process.
• Streamlines the process.
• Minimizes risk of clinical holds.
• Provides regulatory insights.
Consultants provide:
• Expertise in regulatory affairs.
• Guidance tailored to the specific drug characteristics.
• Streamlining of the IND preparation process.
• Prevention of regulatory delays, rejections, or post-submission amendments.
• Increased likelihood of successful IND submission.
Syner-G provides:
• Deep experience in every stage of the IND process.
• Teams of skilled and practiced experts.
• Capabilities around managing the entire product pathway.
• Up to date knowledge of the constantly changing healthcare landscape.