post
Regulatory Medical Writing: Who is your audience?
post
Personalized Medicine Requires a New Approach to Quality Control
post
Empowering and Encouraging Innovation: The Advantages of the FDA’s Orphan Drug Designation for Drug Companies and Patients
post
Overview of FDA Expedited Development and Approval Programs for Serious Conditions
post
The Push for Patient-Focused Drug Development
post
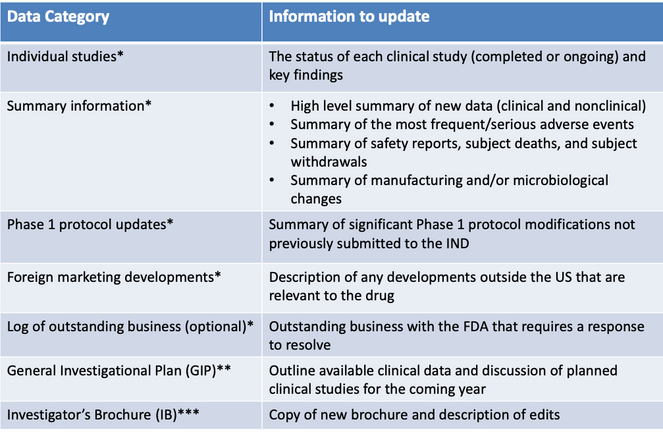
Keep ’em Coming: An Overview of IND Updates
post
From Benchtop to Desktop: 8 Transferable Skills Learned in the Lab that Apply to Medical Writing
post
Statistics in Harmony: The Role of Estimands in Regulatory Writing
post
Current Topics in Orphan Drug Development
post
The Why, What, When, Who, and How of Quality Control (QC) of Medical Writing Deliverables
post
Ready to Submit Your Initial IND?
post
Before you file your IND…
post
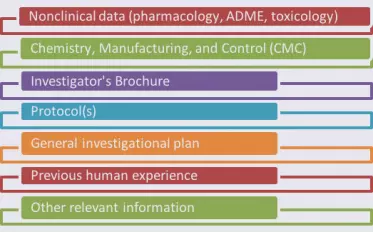
The Ins and Outs of INDs
post
Publishing an eCTD Clinical Study Report: ICH E3
post
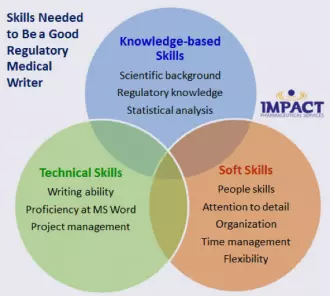
What Skills Do You Need to Be a Good Regulatory Medical Writer?
post
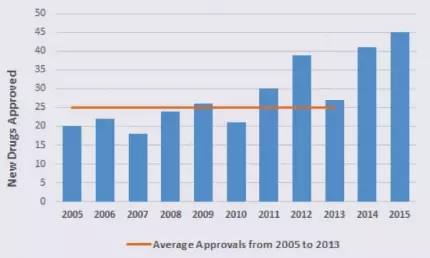
Record Numbers of FDA‑Approved Drugs: Recent Trends
post
Planning Your NDA or BLA Submission: It’s More Than a Gantt Chart!
post
Top Two Things I Learned at DIA RSIDM
post
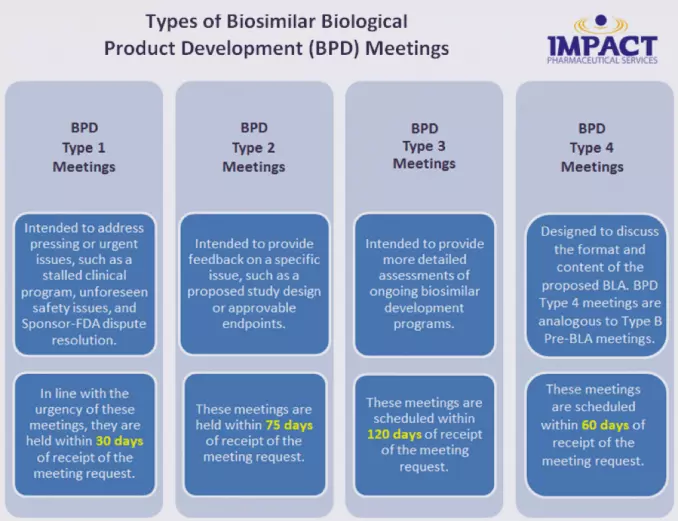
Formal Meetings with FDA for Biosimilar Products
post
How to Make Publishing Clinical Summaries Easier
post
Direct-to-Consumer Advertising of Prescription Drugs