post
Building a QMS (Quality Management System) for Medical Device
post
Manufacturing Gaps in the U.S. Pharmaceutical Industry Exposed by Tariff Wars
post
The Real Cost of Uncertainty: Why On-Again, Off-Again Tariffs Hurt Biotech Supply Chains
post
Accelerating a Cytotoxic Peptide-Drug Conjugate to IND: A Medicinal Chemist’s Journey to IND with Syner-G BioPharma Group
post
Navigating Analytical Method Comparability and Equivalency Under ICH Q14: A New Era for Analytical Procedure Lifecycle Management
post
NextGen Lab Moves: Strategies to Maximize Your Laboratory Relocation Plan
post
Best Practices for System Evaluation and Selection
post
The U.S.-China Trade War and Its Impacts on Investigational Drug Supply Chains
post
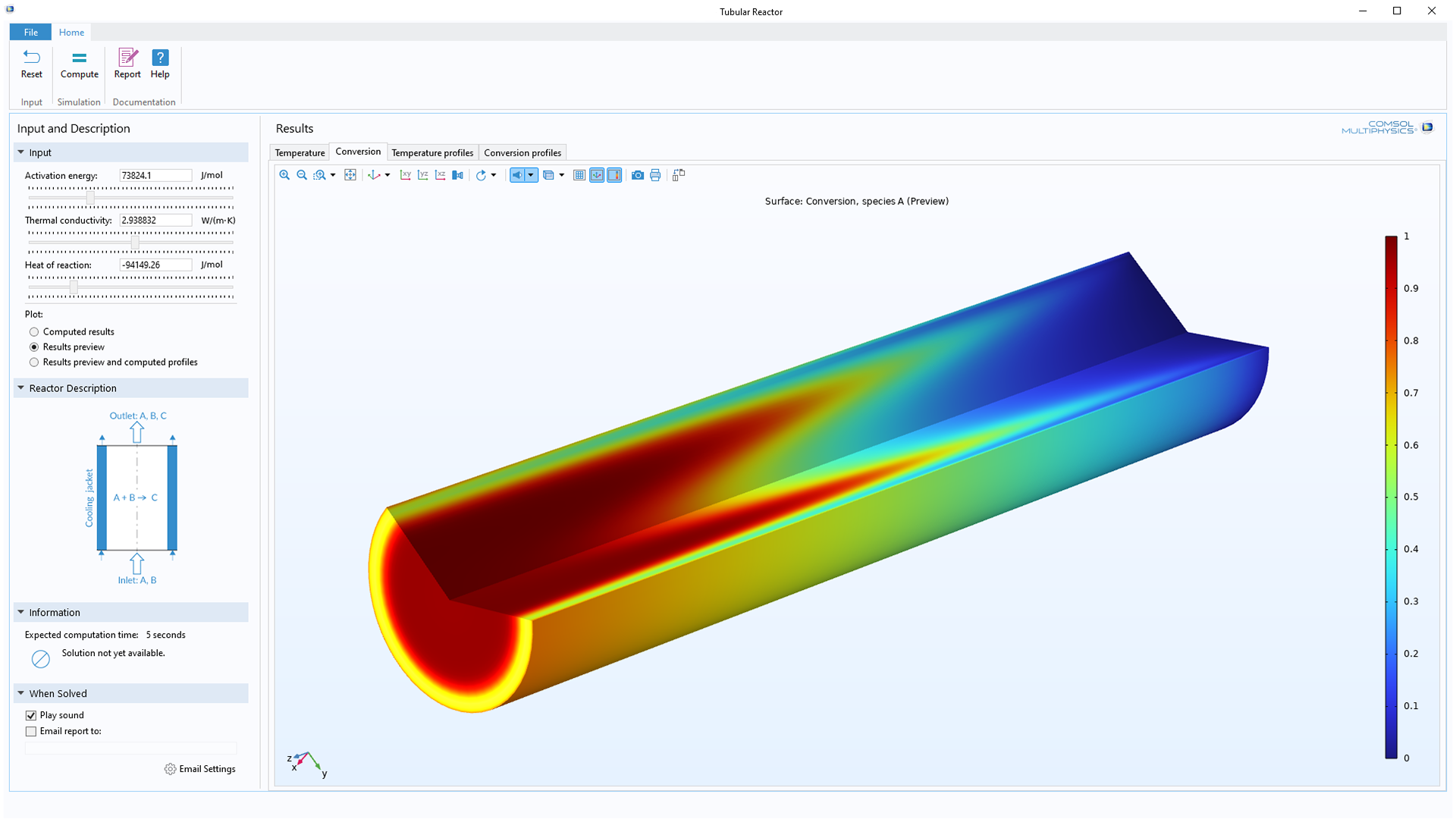
CFD Applications in Design of Manufacturing Processes and Facilities
post
The Evolution of US Biologic Drug Regulations
post
Navigating the Evolution from ICH Q2(R1) to ICH Q2(R2) and Implementation of ICH Q14 in Biopharmaceutical Method Validation
post
Why are Biologics Drugs Different
post
Why Hire an Experienced CMC Consultant for Your Early Phase Studies
post
Understanding the Nuances of a Clinical Study Protocol
post
Exploring Risk-Based Approaches to Raw Material Testing in Pharmaceutical Manufacturing
post
Potency Assurance for Cellular and Gene Therapy Products
post
Conducting Clinical Trials in Australia
post
Syner-G Solutions To Navigating Medical Writing From Home
post
Shortening the Timeline: A Medical Writer’s Guide to Success
post
Writing with Style: Why styles are important in medical writing
post
Prescription for Success: Streamlining Meetings to Craft Compelling Regulatory Documents
post
Syner-G’s Medical Writing Fellowship Program: Preparing Professionals for a Career in Regulatory Medical Writing
post
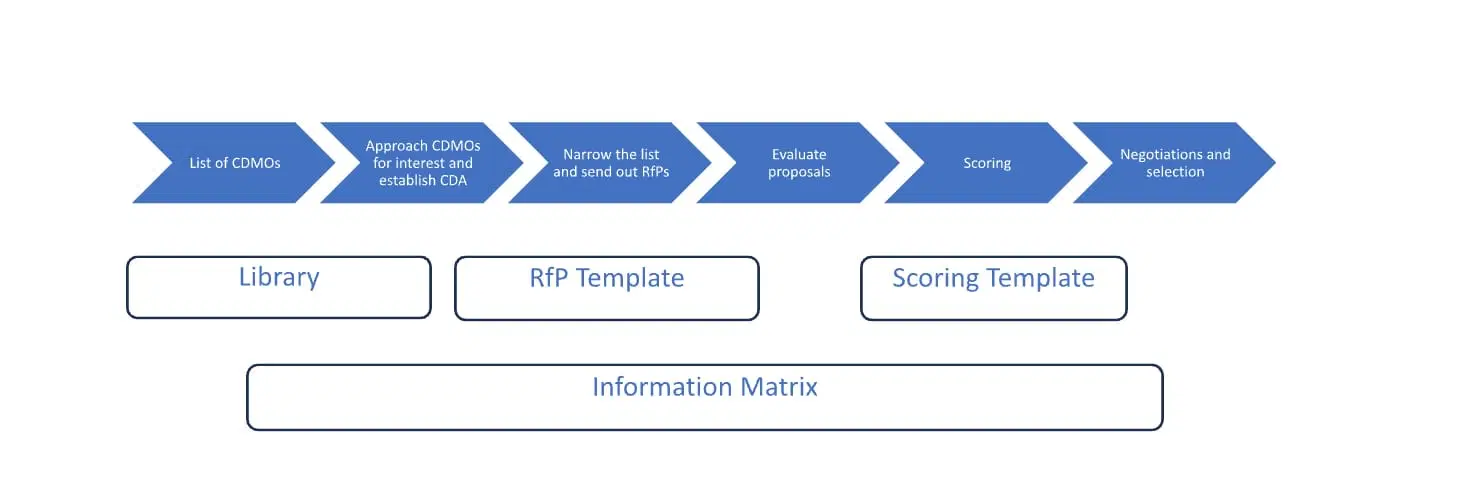
Useful Tools to Facilitate CDMO Selection Process for Biologics
post
Advice for First Time NDA/BLA Submission Teams