The pharmaceutical industry has historically been defined by cautious precision and rigorous scientific methodology. While these traits remain essential for safety, the operational landscape has shifted. In 2026, the pace of innovation is no longer solely dictated by biology and chemistry, but by data and infrastructure.
Cloud and AI in pharma are no longer just emerging trends; they are core enablers of digital transformation. For CIOs, IT leaders, and operations executives, the mandate is clear: modernizing infrastructure and analytics capabilities is a strategic necessity, not merely an IT upgrade.
This article explores how cloud computing and artificial intelligence are reshaping pharmaceutical operations—from R&D and manufacturing to supply chain resilience and regulatory compliance.
From Legacy Systems to Digital Pharma Ecosystems
For decades, pharmaceutical companies relied on on-premise servers and siloed legacy systems. While functional, these environments created fragmented data landscapes where R&D, manufacturing, and regulatory affairs operated in isolation. In a market where speed-to-market and adaptability are paramount, these silos represent a significant bottleneck.
The modern life sciences enterprise requires a holistic view of its operations.
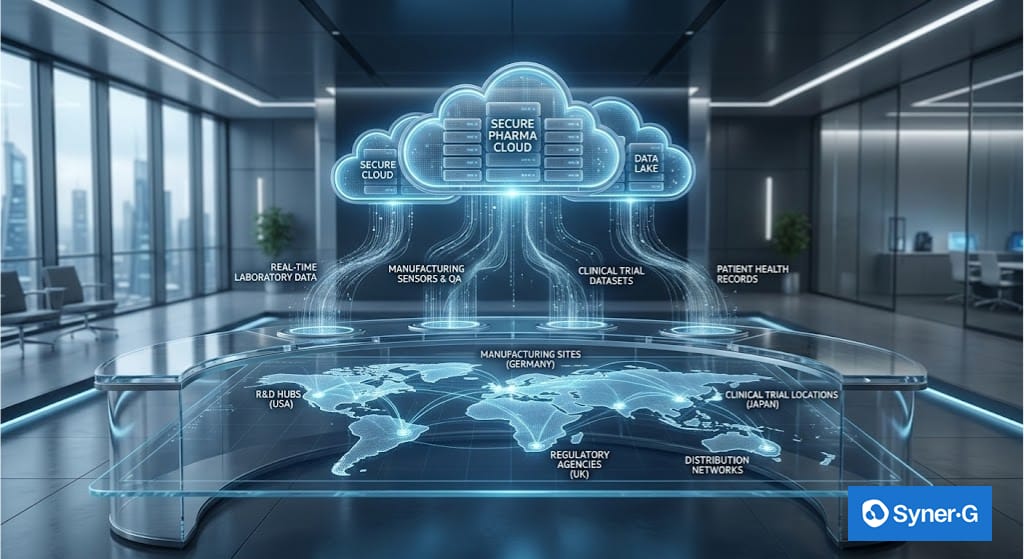
Digital transformation in pharma requires an integrated data ecosystem. By migrating to the cloud, organizations can centralize vast datasets—from genomic sequencing to manufacturing sensor logs—creating a single source of truth. This shift allows for data integration across the life sciences value chain, ensuring that information flows seamlessly from the lab to the patient.
When data is accessible and standardized, it empowers decision-makers to shift from retrospective reporting to real-time operational insight.
Cloud Infrastructure as the Foundation of Pharma Digital Transformation
Cloud computing provides the scalability, flexibility, and reliability necessary to support heavy computational workloads. However, for the pharmaceutical sector, the cloud is more than just storage; it is an environment for collaboration and compliance.
Key benefits of a Cloud-First Strategy include:
- Security & Compliance: Cloud pharma platforms are designed to meet stringent industry requirements, including HIPAA, GxP, and global data privacy standards.
- Global Collaboration: Cloud-native environments allow global teams to collaborate securely on drug development projects without geographical barriers.
- Cost Efficiency: IT leaders can reduce the total cost of ownership regarding infrastructure while simultaneously boosting agility, redirecting capital toward scientific innovation.
Critically, cloud infrastructure is the prerequisite for deploying AI and machine learning in pharma. Without the elastic compute power of the cloud, the complex algorithms required for predictive modeling and simulation simply cannot function at scale.
AI and Machine Learning in Operations
Once data is centralized in the cloud, artificial intelligence (AI) and machine learning (ML) act as the engines of value creation. These technologies are revolutionizing the value chain by automating complex tasks and uncovering patterns invisible to the human eye.
Key applications of AI in pharma operations include:
Accelerated Drug Discovery
AI algorithms can screen millions of molecular structures in a fraction of the time it takes traditional methods, identifying high-potential candidates with greater accuracy.
Optimized Clinical Trials
Machine learning models analyze patient populations and historical data to optimize trial design, improve patient recruitment, and predict site performance. This reduces the time and cost associated with bringing a drug to market.
Predictive Manufacturing and Quality
AI monitors equipment and process parameters in real-time to predict failures before they occur. This ensures continuous production, reduces waste, and improves yield.
Regulatory Intelligence
Natural Language Processing (NLP) scans global regulatory databases to ensure that documentation and submissions remain compliant with evolving standards.
By leveraging data analytics powered by AI, organizations move from reactive problem-solving to proactive optimization.

Digital Transformation of the Pharmaceutical Supply Chain
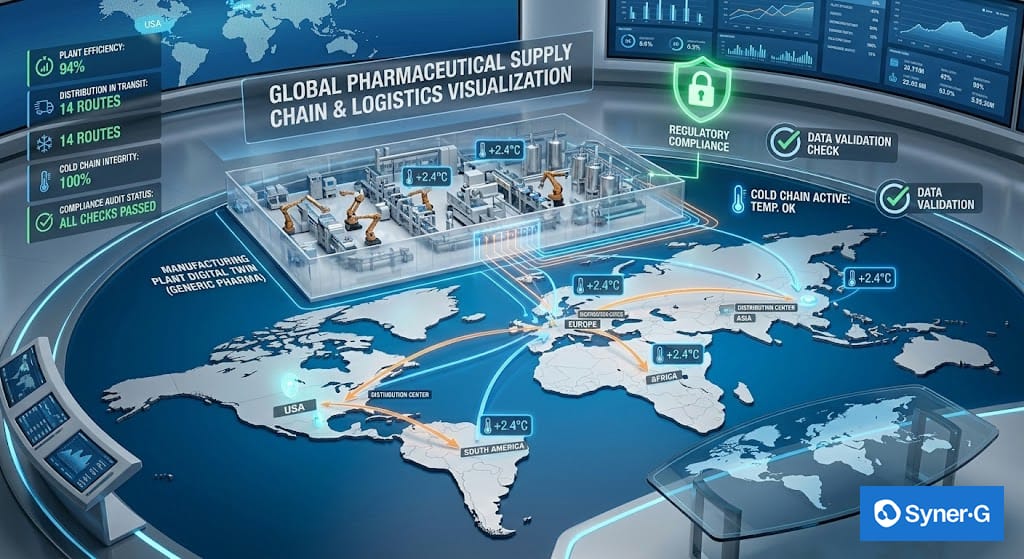
Perhaps nowhere is digital transformation more tangible than in the supply chain. Recent global disruptions highlighted the fragility of traditional logistics models. To build resilience, leaders are turning to digital twins and predictive analytics.
A cloud-enabled digital supply chain provides end-to-end visibility. It allows operations managers to track raw materials, monitor temperature-controlled logistics (cold chain), and predict potential shortages based on geopolitical or environmental factors.
Digital transformation also supports compliance with serialization and traceability requirements, including DSCSA mandates. Integrated digital systems allow unit-level tracking while reducing administrative burden through automated data exchange with ERP platforms.
Data Integrity, Governance, and Regulatory Expectations
As pharma companies embrace these technologies, they must remain cognizant of the regulatory landscape. Regulators (FDA, EMA) are increasingly advocating for data standards, but they also warn that “garbage in, garbage out” is a risk that cannot be taken when patient safety is on the line.
Prioritizing data governance is key to a strong digital strategy, ensuring that data integrity meets the ALCOA+ standard throughout its entire lifecycle:
- Attributable
- Legible
- Contemporaneous
- Original
- Accurate
- (Plus: Complete, Consistent, Enduring, and Available)
By implementing automated data capture and validation tools, companies reduce the risk of human error, ensuring that their digital transformation efforts align with strict compliance standards.
Patient Impact and Personalized Medicine
Ultimately, the goal of digital transformation pharma initiatives is not just operational efficiency; it is better patient care.
When drug discovery is faster, patients get life-saving therapies sooner. When clinical trials are more efficient, participation becomes less burdensome. By harnessing big data, companies can also move toward personalized medicine. Analyzing real-world data and genomic information allows for the development of targeted therapies that offer higher efficacy and fewer side effects for specific patient subgroups.
Strategic Outlook: The Synergy of Tech and Science
The integration of cloud computing and AI represents a strategic inflection point for the life sciences industry. The journey is complex, requiring a balance of technical expertise, scientific knowledge, and regulatory acumen. However, the cost of inaction is far higher.
By adopting these digital tools, your organization does more than just modernize its operations; it positions itself at the forefront of medical innovation. As we look toward the future of healthcare, the synergy between technology and science will define the next era of drug development.
Frequently Asked Questions
How are cloud and AI used in pharma operations?
They support data integration, advanced analytics, predictive manufacturing, supply chain optimization, and regulatory compliance, creating a more agile and data-driven organization.
Why is cloud infrastructure critical for AI in pharma?
AI models require scalable compute power and centralized data environments to function effectively. Cloud platforms provide the necessary elasticity and storage to run these complex algorithms at scale.
What are the regulatory considerations for AI in pharma?
Data integrity, traceability, validation, and governance are essential. Organizations must ensure their digital strategies adhere to ALCOA+ principles to meet FDA and EMA expectations.
The Future of Pharmaceutical Operations
Cloud and AI are driving the next wave of digital transformation in pharmaceutical operations. Companies that invest in integrated, compliant, and analytics-driven platforms will define the future of drug development and delivery.