Bringing a new therapeutic to market is one of the most complex, expensive, and high-stakes endeavors in the modern business world. For biopharmaceutical companies, whether agile startups or established industry giants, the path from molecular discovery to the pharmacy shelf is rarely traveled alone. To navigate the labyrinth of drug development, companies increasingly rely on strategic outsourcing partners.
However, the acronyms governing this industry can be overwhelming. If you are assessing potential partners, you have likely encountered three primary designations: CRO, CMO, and CDMO. While they all play vital roles in the life sciences ecosystem, they serve distinct functions at different stages of the product lifecycle.
Let’s break down these organizations to help you make an informed decision for your company’s future.
What is a CRO?
A Contract Research Organization (CRO) acts as the scientific and administrative engine behind the testing phase of a new drug or medical device. When a pharmaceutical company discovers a potential compound, it cannot simply start selling it; it must prove it is safe and effective through rigorous testing.
CROs specialize in the design, management, and execution of clinical trials. These research organizations provide the deep knowledge required to navigate the ethical and regulatory hurdles of testing on human subjects. While the sponsor company retains ownership of the data and the molecule, the CRO handles the heavy lifting of clinical trial management, including site selection, patient recruitment, and data monitoring.
The value of a CRO lies in its specialized focus. Running a clinical trial requires a massive infrastructure that many biopharmaceutical companies simply do not possess in-house. A CRO bridges this gap by offering services that range from preclinical services to Phase I-IV clinical research. They ensure that the data collected is robust, statistically significant, and compliant with bodies like the FDA or EMA.
However, it is important to note that a standard CRO typically does not manufacture the drug, especially at commercial scale; their primary product is data and regulatory insight. They are the architects of the evidence required to move a therapy forward, serving as the bridge between the laboratory bench and regulatory approval.
What is a CMO?
Once a drug has been formulated and has passed necessary hurdles, or when a company needs test batches for those trials, the focus shifts from research to physical production. This is the domain of the Contract Manufacturing Organization (CMO).
A CMO is primarily focused on contract manufacturing. These organizations possess the facilities, equipment, and technical expertise to produce drug substances and drug products at scale. Biopharmaceutical manufacturing requires incredibly expensive infrastructure, including bioreactors, sterile fill-finish lines, and cleanrooms that meet Good Manufacturing Practice (GMP) standards.
For many bio-pharma companies, building a dedicated manufacturing facility is cost-prohibitive and risky, especially before a drug is approved. A CMO offers a solution by providing manufacturing services on a contract basis. They take the sponsor’s pre-defined formula and process, and they execute it with precision.
The core competencies of a CMO revolve around capacity and consistency. They excel in quality control and quality assurance, ensuring that every pill, vial, or syringe produced meets the exact specifications required for patient safety. Many CMOs focus on executing an existing process rather than designing it, though some offer development support that overlaps with CDMO services.
What is a CDMO?
In recent years, the lines between development and manufacturing have blurred, giving rise to the Contract Development and Manufacturing Organization (CDMO). As the name implies, a CDMO offers a hybrid approach, combining the technical capabilities of contract development with the production power of a manufacturing organization.
A CDMO is designed to partner with pharmaceutical companies much earlier in the lifecycle than a traditional CMO. They often get involved during the pre-clinical or early clinical phases. Their scientists assist with formulation development, process optimization, and stability testing before scaling up for commercial production.
This model is particularly attractive for emerging biotech companies that may have a promising molecule but lack the internal resources to turn that molecule into a stable, deliverable drug. A CDMO handles the “D” (Development)—figuring out how to manufacture the drug efficiently—and the “M” (Manufacturing)—actually producing it.
The primary advantage of the CDMO model is continuity. In a traditional outsourcing model, a company might use one vendor to develop the formulation and a different vendor to manufacture it. This handoff often leads to “tech transfer” risks, where knowledge is lost or processes fail when scaled up. By keeping the development process and commercial manufacturing under one roof, a CDMO minimizes these risks, smoothing the transition from the lab to the plant.
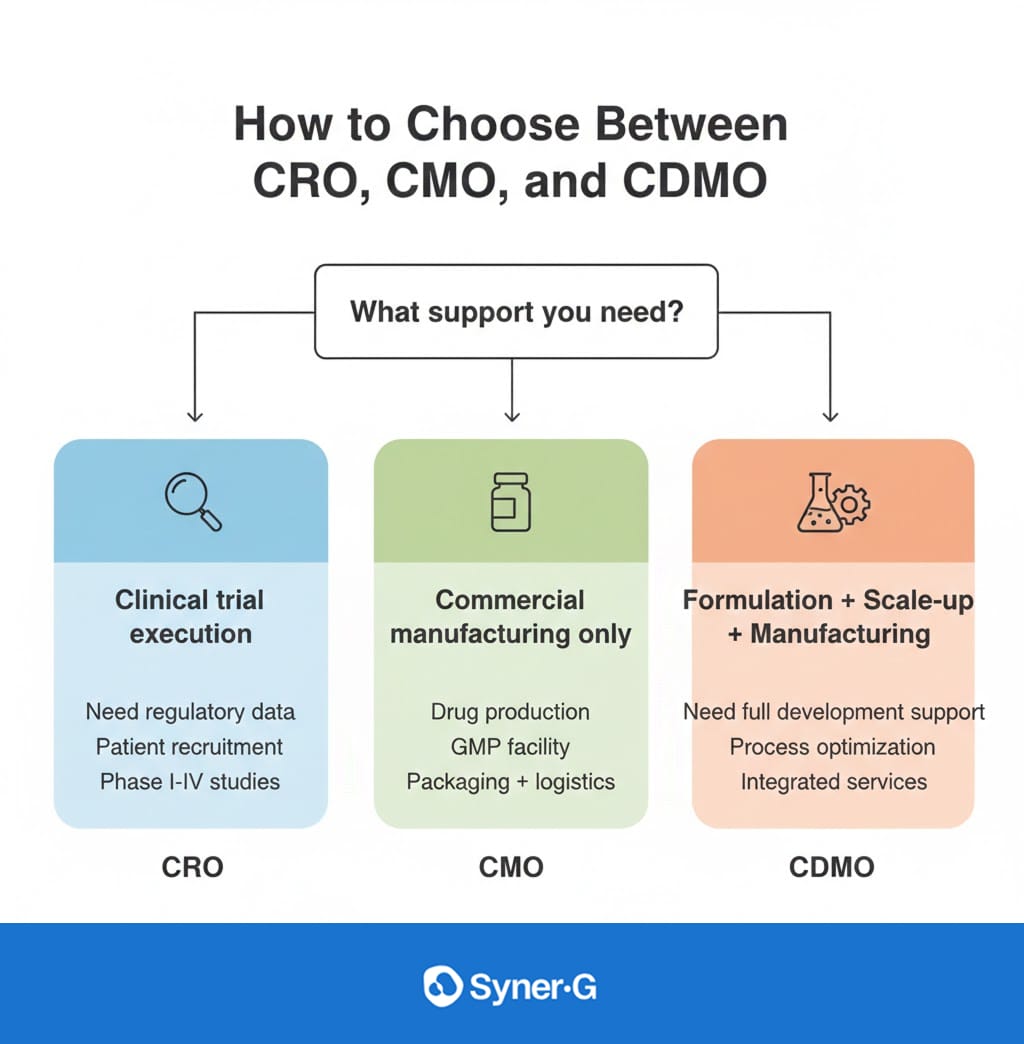
Key Differences at a Glance
To visualize the distinction between CRO, CMO, and CDMO, it is helpful to look at their primary responsibilities and where they fit into your pipeline.
| Feature | CRO (Contract Research Organization) | CMO (Contract Manufacturing Organization) | CDMO (Contract Development & Manufacturing Org) |
| Primary Focus | Focuses on clinical research, trial management, and data collection. They test the drug. | Focuses on large-scale production based on existing processes. They build the drug. | Focuses on both optimizing the drug’s formulation and manufacturing it. They improve and build the drug. |
| Lifecycle Stage | Dominates the clinical phase (Phase I through Phase IV) and regulatory submission. | Enters during late-stage clinical trials for supply and commercial production post-approval. | Can enter as early as pre-clinical development and stay through commercialization. |
| Deliverables | Clinical study reports, regulatory dossiers, and safety data. | Finished drug product or drug substance ready for distribution. | Optimized manufacturing processes, cell lines, and finished drug product. |

How to Choose the Right Partner
Selecting between a CRO, CMO, or CDMO depends entirely on your company’s internal capabilities and current needs. There is no “better” option; there is only the option that fits your business model.
If your internal team consists mostly of scientists who have perfected a molecule but you lack the infrastructure to run a multi-country patient study, you need a CRO. The CRO will handle the logistics of clinical trials while you focus on the science. Conversely, if you have a fully approved drug and simply need more volume to meet market demand, a CMO is the most cost-effective route to scale your manufacturing services.
However, the industry is increasingly trending toward the CDMO model, particularly for complex biologics and cell therapies. If you are a startup with limited CMC (Chemistry, Manufacturing, and Controls) experience, a CDMO can provide the scientific consulting necessary to ensure your drug is actually manufacturable at a commercial scale. They bridge the gap between a concept in a beaker and a product in a bottle.
When evaluating these partners, consider the following actionable criteria:
- Scope of Work: Clearly define if you need data (CRO), pure production capacity (CMO), or process innovation and production (CDMO).
- Regulatory Track Record: Ensure the partner has a history of regulatory compliance with the agencies relevant to your target markets (FDA, EMA, etc.).
- Scale Flexibility: Can the partner handle your needs today (e.g., small clinical batches) and your needs tomorrow (e.g., commercial launch)?
- Technology Transfer: If choosing a CMO, assess how easily you can transfer your formulation development data to their facility. If choosing a CDMO, ask how they handle the internal transition from development to manufacturing.
- Quality Systems: Rigorous quality control and quality assurance protocols are non-negotiable. Audit their facilities to ensure their standards match your own.

Syner-G’s Role in CDMO Partner Selection
As outsourcing models become more complex, many sponsors struggle not just with “CRO vs CMO vs CDMO,” but with picking the right specific partner for their molecule and modality.
Syner‑G focuses on this decision point, helping biopharma companies evaluate, select, and manage CDMOs so they can move from concept to commercial supply with fewer surprises.
Instead of simply handing you a list of vendors, Syner‑G brings structured decision frameworks, risk‑based assessments, and deep CMC expertise to your CDMO selection process. The team assesses technical fit, regulatory track record, quality systems, capacity, and long‑term scalability, then helps you negotiate realistic scopes and governance models that align with your strategy.
For emerging biotechs in particular, Syner‑G acts as an extension of your internal team—translating your scientific and business needs into clear requirements for development and manufacturing partners. This ensures that the CDMO you choose is not just capable of producing material but is also equipped to support your asset across phases, manage tech transfer efficiently, and scale with your pipeline.
Moving Forward with Confidence
The pharmaceutical industry is built on collaboration. Whether you engage a CRO to manage your clinical research, a CMO to handle your supply chain, or a CDMO to guide your development process from start to finish, the goal remains the same: delivering life-saving therapies to patients safely and efficiently.
By understanding the distinct roles of CRO vs CMO vs CDMO, you can build a supply chain strategy that is robust, compliant, and scalable. Take the time to vet your potential partners thoroughly. In this industry, your partners are an extension of your own company, and their expertise is the catalyst that turns scientific potential into medical reality.





